 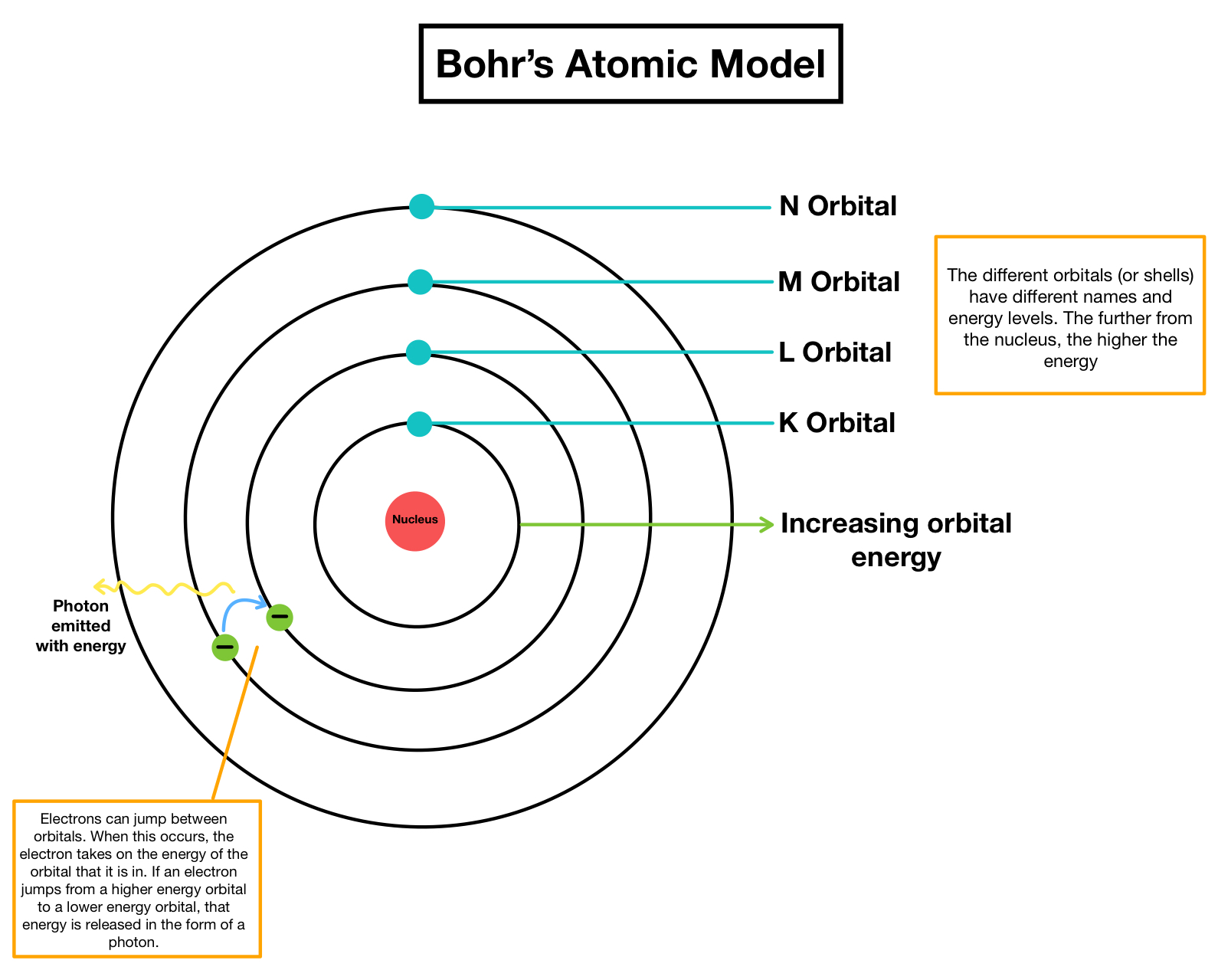
However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner.\), the lines in this series correspond to transitions from higher-energy orbits (n > 2) to the second orbit (n = 2). Niels Bohr’s most-noted contributions to contemporary thought center around his work with quantum theory. This model incorporated Rutherfords planetary model and explained the atomic spectrum of hydrogen. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. Niels Bohr made significant contributions to atomic theory by proposing a model of the atom in 1913 called the Bohrs Model. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. To overcome this, in 1921, Rutherford with the help of Niels Bohr put forth a theory, hypothesizing the existence of a neutral-charged particle that had the same mass of a proton. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thus, the Bohr model for heavier atoms described electron shells. Bohr model, description of the structure of atoms proposed in 1913 by the Danish physicist Niels Bohr. 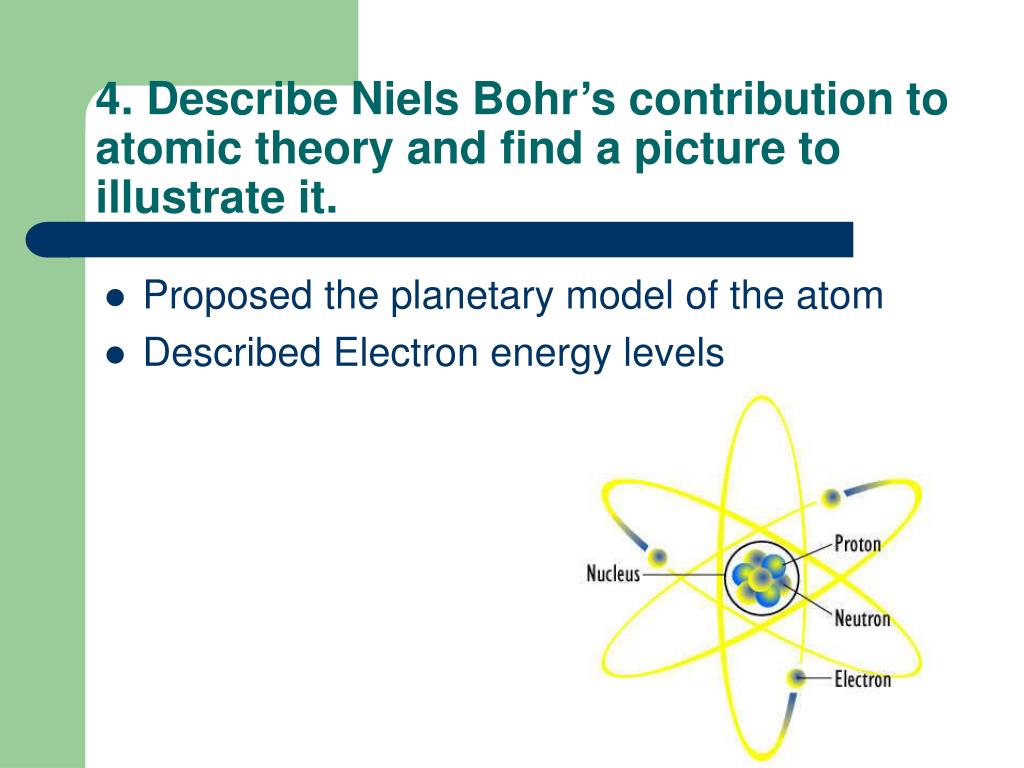
In particular, he developed the Bohr model of the atom (and later the liquid drop model) and the principles of correspondence and complementarity. Once the level was full, additional electrons would be bumped up to the next level. Niels Bohr was a Danish physicist who made fundamental contributions to understanding the structure of atoms and to the early development of quantum mechanics. He won the 1922 Nobel Prize in physics for his ideas and years later, after working on the Manhattan Project, called for peaceful applications of atomic energy. Bohr believed each electron orbit could only hold a set number of electrons. Niels Bohr was a Nobel Prize-winning physicist and humanitarian who came up with a revolutionary theory on atomic structures and radiation emission. More electrons were required to cancel out the positive charge of all of these protons. Because the ratios were the same, Dalton was able to say that atoms of different elements combine in whole number ratios. Niels Bohr was a Danish physicist who had a theory about atoms that he called the atomic model. 
Entitled On the Constitution of Atoms and Molecules, the article brought together for. Thompson, Ernest Rutherford, Niels Bohr, James Chadwick and Ernest Schrodinger each contributed greatly to the modern atomic theory by finding the actual evidence. In 1913, Niels Bohr published a groundbreaking paper that introduced a new way of understanding atomic phenomena ( 1 ). Heavier atoms contain more protons in the nucleus than the hydrogen atom. One hundred years ago, Niels Bohrs pioneering paper on the electronic structure of the hydrogen atom revolutionized atomic theory. Bohr developed a model of the atom that predicts how the atom will behave.Therefore, option (A) is correct.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
